Повышение продовольственной безопасности с помощью устойчивых биотехнологий: ориентация на нанокапсулирование
Повышение продовольственной безопасности с помощью устойчивых биотехнологий: ориентация на нанокапсулирование
Аннотация
Предпосылки: Мировая продовольственная система сталкивается с давлением ввиду изменения климата, уязвимости цепочек поставок и недостаточного питания. На фоне этих вызовов нанокапсулирование стало перспективным подходом, позволяющим защищать биологически активные соединения, повышать их биодоступность и обеспечивать надежную поставку в сельском хозяйстве.
Цель и методология: В данном систематическом обзоре анализируется состояние технологий нанокапсулирования (2018–2025 гг.) и их роль в укреплении четырех основ продовольственной безопасности (наличие, доступность, использование и стабильность). С использованием руководящих принципов PRISMA 2020 в ходе систематического поиска по пяти базам данных (Scopus, Web of Science, PubMed, ScienceDirect и Google Scholar) было выявлено 50 подходящих рецензируемых исследований. Данные были обобщены с учетом типа носителя, физико-химических свойств и области применения.
Результаты и выводы: Анализ демонстрирует преобладание полимерных и липидных наноносителей (30–500 нм), разработанных для трех основных направлений применения: экологически безопасная доставка агрохимикатов, обогащение продуктов микроэлементами и создание функциональной пищевой упаковки. Несмотря на доказанную техническую эффективность, массовое внедрение по-прежнему остается проблемой. Нанокапсулирование преобразуется из экспериментальной новинки в жизнеспособное решение в области продовольственной безопасности. Однако его широкое внедрение в настоящее время ограничивается фрагментацией нормативной базы и опасениями потребителей по поводу безопасности. В будущем необходимо уделять особое внимание согласованию протоколов безопасности и «зеленым» методам синтеза, чтобы обеспечить безопасность и устойчивость данных технологий.
1. Introduction
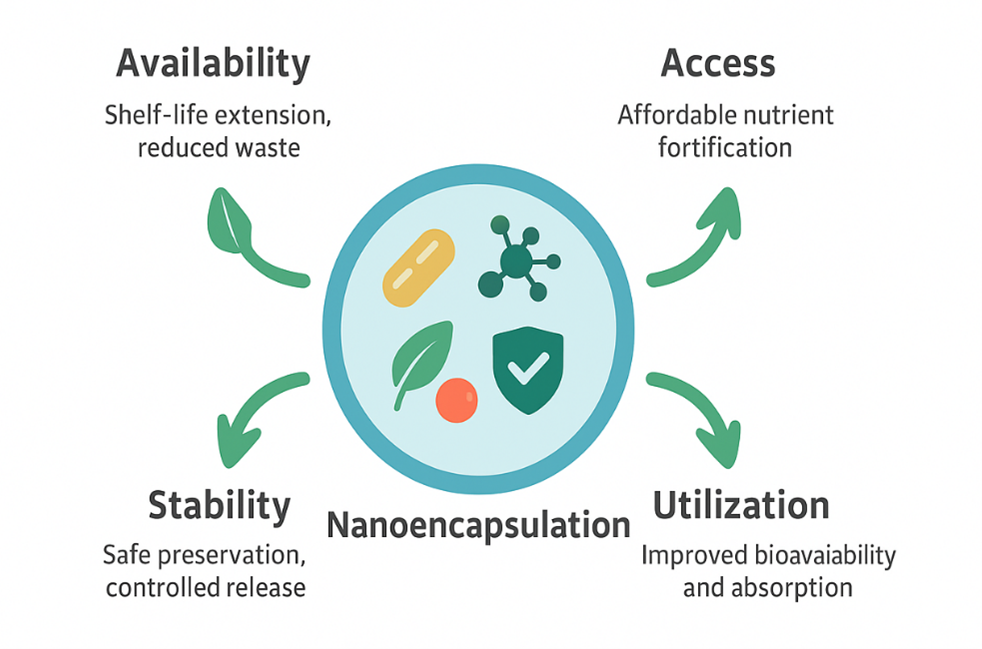
Figure 1 - Graphic Abstract
2. Nanoencapsulation (principle, classification, and delivery methods)
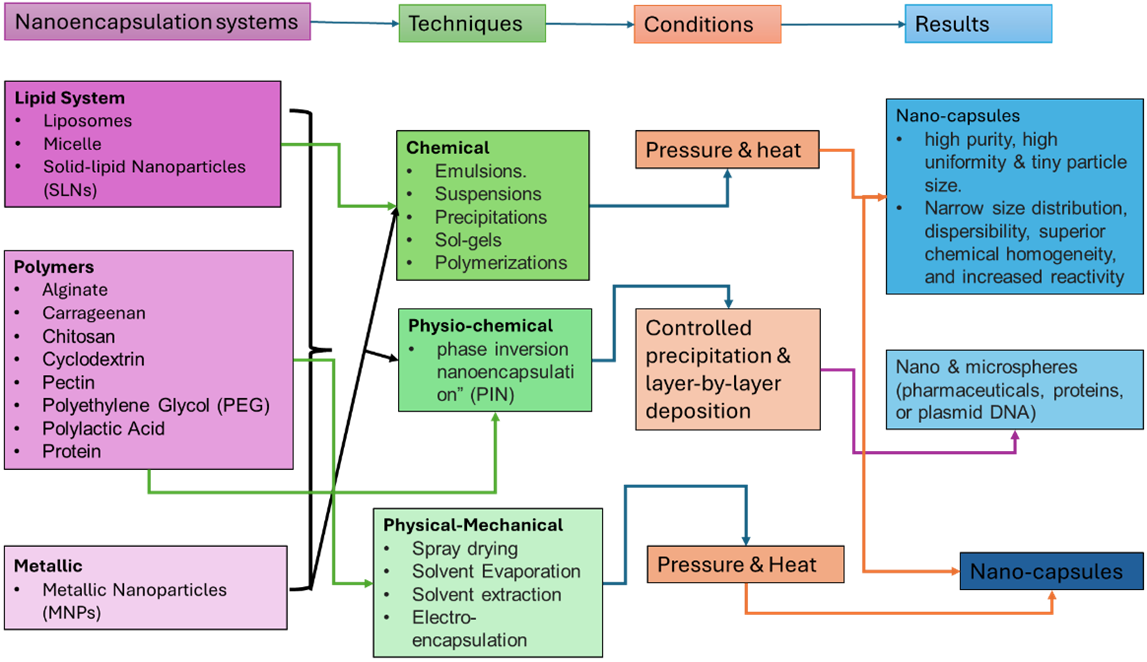
Figure 2 - A summative scheme showing the different ways of forming nanoscale capsules, the techniques involved, the different conditions required, and the resulting nanoscale carrier
At its core, nanoencapsulation operates on the principle of isolating functional compounds within engineered carrier matrices that act as physical and chemical shields
. These carriers are designed to enhance stability, modulate release kinetics, and improve interaction with biological systems. The fabrication of nanocarriers involves a range of chemical, physicochemical, and mechanical methods, resulting in distinct nanostructures with tailored functional properties (Figure 1). Collectively, these approaches significantly enhance the bioavailability and functional efficacy of encapsulated compounds . For the purposes of this review, the term "nanoencapsulation" refers to delivery systems with characteristic dimensions ranging from 1 to 1000 nm. This extended submicron definition is adopted because many food‑grade colloidal systems, polymeric complexes, and lipid assemblies exhibit their most relevant functional, stability‑enhancing, and bioavailability‑related properties within this broader nanoscale range. Such systems remain consistent with regulatory and toxicological frameworks applied to food nanotechnology while capturing the practical realities of industrial food formulations. Nanoencapsulation is a sophisticated technological process involving the entrapment of sensitive bioactive molecules such as vitamins, polyphenols, essential oils, antioxidants, and probiotics within nanoscale carrier systems , , . The primary functional advantage of this approach lies in its ability to protect labile compounds from physicochemical degradation during food processing, storage, transport, and gastrointestinal digestion . By forming a protective barrier around the encapsulated core, nanoencapsulation minimizes losses caused by exposure to oxygen, light, moisture, and temperature fluctuations, while simultaneously enabling controlled and targeted release . Importantly, the scope of nanoencapsulation extends beyond nutrient protection alone. Its applications directly contribute to the four pillars of food security defined by the Food and Agriculture Organization (FAO): availability, access, utilization, and stability . Through shelf-life extension, nutrient fortification, improved absorption, and precision delivery in agriculture, nanoencapsulation represents a convergent technology capable of addressing multiple dimensions of food security simultaneously. The selection of encapsulating materials is governed by biocompatibility, biodegradability, food safety, and the ability to form stable carrier structures . Based on current evidence, food‑grade nanocarriers can be broadly classified into four major material categories:Biodegradable Polymers: Natural biopolymers such as chitosan, starch, alginate, and gelatin are among the most widely investigated materials for nanoencapsulation. These polymers exhibit high encapsulation efficiency, tunable surface properties, and environmentally benign degradation pathways
. Advances in polymer chemistry have enabled targeted delivery applications, with increasing relevance for both food systems and biomedical nutrition .Lipid‑Based Systems: Lipid nanocarriers are particularly effective for the delivery of hydrophobic compounds
. These systems include liposomes, solid lipid nanoparticles (SLNs), and nanostructured lipid carriers (NLCs) . A critical distinction must be made between nanoemulsions, which are liquid‑in‑liquid colloidal dispersions, and solid lipid nanoparticles, as their physicochemical behavior, toxicological profiles, and regulatory requirements differ substantially.Protein‑Based Carriers: Food proteins such as casein, whey proteins, and plant‑derived proteins
serve as natural delivery vehicles with high nutritional value and adaptable structural properties . Their ability to self‑assemble into nanoscale structures enables precision delivery and controlled digestion within the gastrointestinal tract .Carbohydrate‑Based Systems: Sustainable carbohydrate materials, including pectin, cellulose, and dextran
, are increasingly used to form robust matrices for antimicrobial and antioxidant delivery, particularly in active food packaging applications . Beyond material selection, the efficacy of nanoencapsulation systems is strongly influenced by the mechanism through which bioactives are released. Contemporary research increasingly emphasizes multi‑modal release pathways , including diffusion through the carrier matrix, erosion of the encapsulating shell, swelling‑induced pore expansion, and enzymatic degradation triggered by gastrointestinal conditions .3. Review Methodology
The systematic review was conducted in accordance with PRISMA 2020 guidelines
, ensuring transparency, reproducibility, and methodological rigor.Databases & Search Period: Literature published between January 2018, and December 2025 was retrieved from Scopus, Web of Science, PubMed, ScienceDirect, and Google Scholar.
Search Strategy: Structured Boolean queries were applied, including controlled vocabulary (MeSH terms where applicable). Reference lists of selected articles were also screened (snowballing). Search terms included “nanoencapsulation,” “food security,” “nanocarriers,” and “bioavailability.”
Eligibility Criteria (Inclusion and exclusion): Inclusion criteria were emphasized on peer-reviewed experimental studies, systematic reviews, and meta-analyses reporting quantitative performance metrics (e.g., encapsulation efficiency, release kinetics, bioavailability), while the exclusion criteria were focused on grey literature, conference abstracts, non-English articles, and studies lacking physicochemical or biological performance data.
Screening Process: Two-stage screening (title/abstract to full text) was performed independently, with discrepancies resolved by consensus.
Data Extraction & Synthesis: Extracted variables included nanocarrier composition, size (≤1000 nm), encapsulation efficiency, stability, release behavior, application domain, and safety/toxicological evidence. Results were synthesized narratively and thematically.
Risk of Bias Assessment: Methodological quality was evaluated using adapted criteria from SYRCLE and Cochrane
frameworks for experimental studies.4. Results
A total of 50 studies published between 2018 and 2025 were identified for inclusion. As detailed in the PRISMA flow diagram (Figure 2), these studies were thematically categorized into seven major domains, with a dominant emphasis on bioavailability enhancement, sustainable agriculture, and active packaging. Polymeric and lipid-based nanocarriers were the most frequently reported systems. The particle sizes predominantly ranged between 30 nm and 500 nm, a range that balances cellular uptake efficiency with physical stability in food matrices. The following table summarizes key experimental studies, categorized by their primary application domain.
Table 1 - Summary of recent nanoencapsulation studies (2018-2025) categorized by application domain
Application Domain | Nanocarrier Type | Size (nm) | Key Performance Indicators | Specific Application | Reference |
Sustainable Agriculture | Biopolymer nanoparticles | 100–400 | Encapsulation efficiency, yield response | Crop biostimulants | |
Nano-agrochemicals | 50–300 | Controlled release kinetics | Sustainable agriculture | ||
Nanofertilizers | 30–200 | Nutrient release kinetics | Precision farming | ||
Chitosan nanoparticles | 80–250 | Uptake efficiency | Crop nutrition | ||
Bioavailability & Fortification | Nanoemulsions | 50–300 | Encapsulation efficiency | Food fortification | |
Lipid nanocarriers | 80–200 | Bioaccessibility | Micronutrient delivery | ||
Protein-lipid carriers | 100–500 | Bioavailability enhancement | Plant-based foods | ||
Solid lipid nanoparticles | 90–250 | Sustained release | Vitamin delivery | ||
Active Packaging & Preservation | Nanocomposite films | - | Mechanical strength | Active packaging | |
Biopolymer films | - | Antimicrobial release | Smart packaging | ||
AgNP packaging | - | Migration rate | Food contact materials | ||
Essential oil nanoemulsions | 30–150 | Antifungal activity | Food preservation | ||
Food Safety & Regulation | Metal nanoparticles | 10–100 | Cytotoxicity assessment | Food safety toxicology | |
Nanoencapsulated vitamins | 100–300 | Dose-response relationships | Toxicology | ||
Food-grade nanomaterials | - | Risk thresholds | Regulatory framework | , | |
Polymeric nanocapsules | 50–200 | Mycotoxin inhibition | Grain storage safety | ||
Green & Functional Carriers | Polysaccharide matrices | 150–600 | Stability under stress | Functional foods | |
Nanoemulsions | 40–200 | Shelf-life extension | Green food safety | ||
Polymer nanoparticles | 70–300 | Volatility reduction | Essential oil delivery |
As shown above, several studies consistently demonstrated that nanocarriers, particularly chitosan and biopolymer-based systems, significantly improve the delivery of agrochemicals. Khan et al. (2022) highlighted that reducing particle size to the 50–250 nm range enhances uptake efficiency in crops, reducing the total amount of active ingredients required. Research by Zhang et al. (2021) and McClements (2020) confirmed that these carriers protect sensitive compounds from gastric degradation, thereby increasing bioaccessibility in the intestinal tract. Recent trends show a shift toward nanocomposite films that not only act as barriers but also release antimicrobial agents; however, safety studies emphasize the need to monitor them for cytotoxicity, and regulatory frameworks
are still evolving to establish clear risk thresholds for chronic exposure .5. Discussion and Future Directions
While nanoencapsulation offers a promising future for boosting food security, transitioning from laboratory innovation to mainstream food systems requires systematic mitigation of safety, regulatory, and social barriers
.Nanotoxicology and Human Health Safety: The very properties that make nanocarriers effective small particle size, high surface area, and the ability to cross biological barriers also underpin their potential risks. Concerns have been raised regarding the bioaccumulation and long-term health outcomes of chronic exposure to nanomaterials in the diet
. Recent research by Mahajan et al. (2025) emphasizes that nanotoxicological assessments must become an essential prerequisite for commercialization, utilizing both in vitro and in vivo models to evaluate the cytotoxicity and genotoxicity (the potential for nanocarriers to damage cellular structures or DNA), immunological Impacts (how these materials interact with the body’s immune response ), and gut Microbiome Interaction (investigating how nanocarriers influence the diversity and function of intestinal microbiota), which are critical factors in overall metabolic health . It should also be noted that factors such as surface chemistry, biodegradation rates, and particle size are primary determinants of toxicity and environmental persistence.The Global Regulatory Landscape: Currently, there is a lack of international harmonization in how nano-enabled food products are governed
. Regulatory bodies are adopting cautious, evolving positions; the EFSA (European Food Safety Authority) enforces mandatory pre-market approval for engineered nanomaterials. Producers must submit comprehensive dossiers covering toxicokinetic reports, exposure levels, and allergenicity . The U.S. FDA (Food and Drug Administration) generally evaluates these ingredients within the Generally Recognized as Safe (GRAS) framework but applies heightened scrutiny to particle size and stability . And the FAO/WHO have called for harmonized international standards and transparent safety protocols . Such frameworks are necessary to facilitate safe international trade and ensure that “nano” claims are backed by standardized testing methods and traceability.Transparency and Consumer Perception: Consumer acceptance remains one of the most significant barriers to adoption . Public attitudes are often shaped by a tension between perceived risks and benefits. These tend to create what is known as the “Nano-Fear” factor, a gap that causes many consumers to equate or symbolize it with the term “nano” (artificiality or unknown) risks
, . Acceptance Drivers which are more localized; this has been evidenced in recent surveys, which indicated that consumers are more likely to accept the technology when it is linked to tangible benefits , such as nutrient fortification or reduced food waste, and when products are transparently labeled . Overcoming these gaps requires participatory risk, communication, consumer education, and potentially a shift in labeling nomenclature to earn trust.5.1. Future Directions
The transition of nano-based foods from the Lab to Market beyond the current experimental stages, future research and policy must prioritize:
Enhancement of safety protocols by developing standardized, long-term toxicological protocols that will specifically account for nano-bio interactions and environmental persistence
.Tech-Innovations that lead to the development of smart and stimuli-responsive nano-systems, which allow the integration of biosensing with targeted delivery
, . These high-tech nano-systems will allow “on-demand” release in response to physiological signals like pH or enzyme activity , , , .Integrating the concept of a circular economy, which is believed to be a good strategy because of its conceptual shift (waste-to-resource) strategy
, by using plant-derived biopolymers and materials from agro-industrial waste valorization .Harmonization of global regulatory bodies on the concept of nano-based practices: this will allow coordinated guidelines (EFSA, FDA, and FAO/WHO), which are essential to reduce duplicative assessments and provide a “gold standard” for global governance
, , .By systematically addressing these and other technical and ethical challenges, nanoencapsulation can evolve into a mainstream enabler of resilient and equitable food systems, directly contributing to UN Sustainable Development Goals: Zero Hunger (SDG 2), Good Health and Well-being (SDG 3), and Responsible Consumption and Production (SDG 12).
6. Conclusion
This review assessed and confirmed that nanoencapsulation represents a transformative strategic frontier in sustainable food systems (food biotechnology). By systematically analyzing recent research and literature, it is evident that nanocarriers, particularly biodegradable polymers and lipid systems, offer robust solutions for stabilizing volatile nutrients and reducing agricultural chemical runoff. This technology effectively addresses the “utilization” and “stability” dimensions of food security by extending shelf life and enhancing the bioavailability of functional foods. However, the transition from laboratory efficacy to industrial application and finally to consumer acceptance is currently stalled by the “nano-fear” factor gap and a lack of harmonized global regulation by regulatory bodies. To fully explore the potential of nano-based food technology, future research must rotate from characterization-based studies to long-term toxicological assessments and circular-economy manufacturing. And finally, for the nano-based food technology to serve as a strong pillar of resilient global food systems, it must be developed within a framework that balances technological innovation with rigorous safety standards and transparent consumer communication.
